The vascular supply to the head and neck is chiefly derived from the carotid system of arteries. Numerous variations have been reported regarding the level of origin of the external carotid artery and its branching pattern. Clinically, the branches of the external carotid artery are important in head and neck surgeries involving the thyroid, oro-facial reconstruction, diagnosis and treatment of tumors of the head and neck. This study aimed to observe the origin and branching pattern of the external carotid artery in the Indian Punjabi population. The study was done on both sides of 15 formalin-fixed adult human cadavers, thus making up to 30 carotid systems. The level of origin of the external carotid artery and its individual branches were measured. Out of the 30 carotid systems studied, only four showed the typical level of origin at the upper border of the thyroid cartilage, with trifurcations seen in eight cases and one case showing tetrafurcation. 53.33% of the external carotid systems showed the presence of at least one common trunk of origin, of which linguo-facial trunk was seen in 26.67%, pharyngo- occipital trunk in 23.33% and auriculo-occipital trunk in 13.33% cases. Moreover, an aberrant ascending pharyngeal artery was noticed, with communication to the internal carotid artery.
Origin and branching pattern of external carotid artery – a cadaveric study
Aby S. Charles 1, Suganthy Rabi 2, Anjali Jain 3, Parminder K. Rana 4
1 Christian Medical College, Ludhiana*
2 Christian Medical College, Vellore
3 Christian Medical College, Ludhiana
4 Christian Medical College, Ludhiana**
Present affiliation:
* Christian Medical College, Vellore
** Adelaide Medical School, Adelaide
SUMMARY
Sign up or Login
INTRODUCTION
The vascular supply to the head and neck is chiefly derived from the carotid system of arteries. Additional supply comes from the branches of the subclavian artery, most importantly the vertebral artery. The carotid system of arteries begins as the common carotid artery (CCA), which bifurcates into the external carotid artery (ECA) and the internal carotid artery (ICA) (Mata et al., 2012; Hollinshead et al., 1968). The level of carotid bifurcation cannot be predicted by any known clinical sign. Most of the literature states that bifurcation of the common carotid artery occurs at the level of the superior border of the thyroid cartilage (Lo et al., 2006).
The ECA has eight branches, namely the superior thyroid artery, the ascending pharyngeal artery, the lingual artery, the facial artery, the occipital artery, the posterior auricular artery, the superficial temporal artery and the maxillary artery (Hollinshead et al., 1968). A good knowledge of the normal anatomy and variations of ECA and its branches are of prime importance in both surgical and medical specialties. Both carotid endarterectomy and carotid stenting to prevent recurrence of stroke warrant a thorough knowledge of the carotid system (Rajamani and Chaturvedi, 2011). Extracranial-intracranial bypass procedure for revascularization uses the ECA or one of its branches as donor vessels (Germans and Regli, 2014). Radiologists must have a good preexisting knowledge of the external carotid arterial system and expected variations in order to identify the various arteries. This applies specially to tests like CT angiography, which are done prior to major surgeries (Acar et al., 2013). Hence, this study aims at observing the origin and branching pattern of the external carotid artery in the Indian Punjabi population.
MATERIALS AND METHODS
Ethical approval was obtained from the Institutional Review Board of the Christian Medical College, Ludhiana, where the study was conducted. Fifteen formalin-fixed adult cadavers of unknown age, ten male and five females, were obtained from the Department of Anatomy of the institution. These cadavers were being used by medical students as part of their training and was obtained through a body donation program. Bilateral dissection was done. Thus, 30 ECAs were studied for their origin and branching pattern. Cadavers with any obvious congenital anomaly of the head and neck region or those in which the carotid system was used for embalming purposes were excluded from the study.
The cadaver was dissected in a supine position with the neck extended. The skin and superficial fascia containing platysma with nerves and the deep fascia were flected. The boundaries of the carotid triangle were ascertained. The inferior attachment of the sternocleidomastoid muscle was detached to better expose the area. Fat and fascia from the area between the posterior belly of the digastric and the superior belly of the omohyoid were removed, exposing the CCA and ICA medially and the internal jugular vein laterally. The ECA was identified lying anteromedial to the ICA. The hypoglossal nerve was identified and followed forwards across the ECA. Ansa cervicalis was identified and removed. The posterior belly of the digastric, stylohyoid, and thyrohyoid muscles was reflected. The carotid sheath was dissected to expose the CCA and its bifurcation into ICA and ECA. The ECA was traced in the digastric triangle. The hypoglossal nerve was preserved to serve as landmark.
In the parotid gland, the ECA was traced after removing the gland piecemeal. The termination of the ECA was studied by division of the zygomatic arch and removal of part of the mandible along with the attached muscles. The branches of the ECA were identified and followed to confirm their identity and course. This was verified by a second investigator.
The level of bifurcation of the CCA was noted in relation to the highest point of the upper border of the thyroid cartilage with a scale, and then this distance was measured using Vernier calipers. The distance of origin of the individual branches of the ECA from the point of carotid bifurcation was measured along the tortuosity of the vessel using an inelastic thread and measured by the Vernier calipers. The variations in the branching pattern, if any, were documented. Descriptive statistics were used and analyzed using SPSS Version 23.
RESULTS
Carotid bifurcation
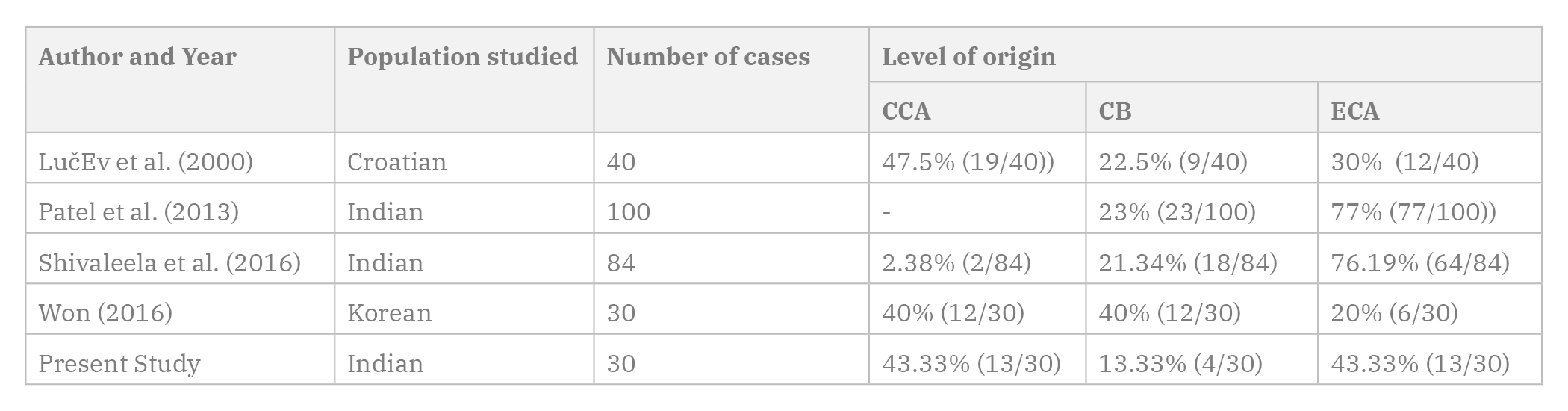
In the present study, the carotid bifurcation was seen at a mean distance of 1.28±0.93 cm above the level of the upper border of the thyroid cartilage in 25 out of 30 (83.33%) cases, out of which 11 were on the left side, and 14 on the right side (Fig. 1A). The eleven cases on the left were at a mean distance of 1.55±0.96 cm, while the 14 cases on the right were at a mean distance of 1.06±0.86 cm from the upper border of the thyroid cartilage. Only in four out of 30 (13.33%) cases, the carotid bifurcation occurred at the level of the upper border of the thyroid cartilage, three cases on the left and one case on the right side. In one case (3.33%), on the left side, the bifurcation occurred 1.2 cm below the level of the upper border of the thyroid cartilage (Fig. 1B).
In the present study, the branches of ECA took origin not only from the ECA, but also from the CCA or at the level of bifurcation. A trifurcation, meaning a branch arising from the carotid bifurcation, was noted in eight cases. In three cases, the superior thyroid artery took origin from the carotid bifurcation; in four cases, the ascending pharyngeal artery took origin from the carotid bifurcation, and in one case the occipital artery took origin at the level of the carotid bifurcation (Fig. 2A). One case of tetrafurcation was observed, where both the superior thyroid artery and the ascending pharyngeal artery took origin from the carotid bifurcation.
Of the 30 cases studied, in 13 cases the superior thyroid artery took origin from the CCA.
In one case, on the left side, the ascending pharyngeal artery took origin from the CCA, after which it travelled upwards and medially along with the ICA. In this particular case, in addition to the ascending pharyngeal artery, a linguo-facial trunk arose from the CCA. At a distance of 1.2 cm from its origin, the artery communicated with the ICA by a very short communicating branch, before continuing on its course. The ICA gave off two branches in the neck just above this level. On following these branches, they appeared to be entering the cranial cavity at the base of the skull (Fig. 2B).
Length of ECA
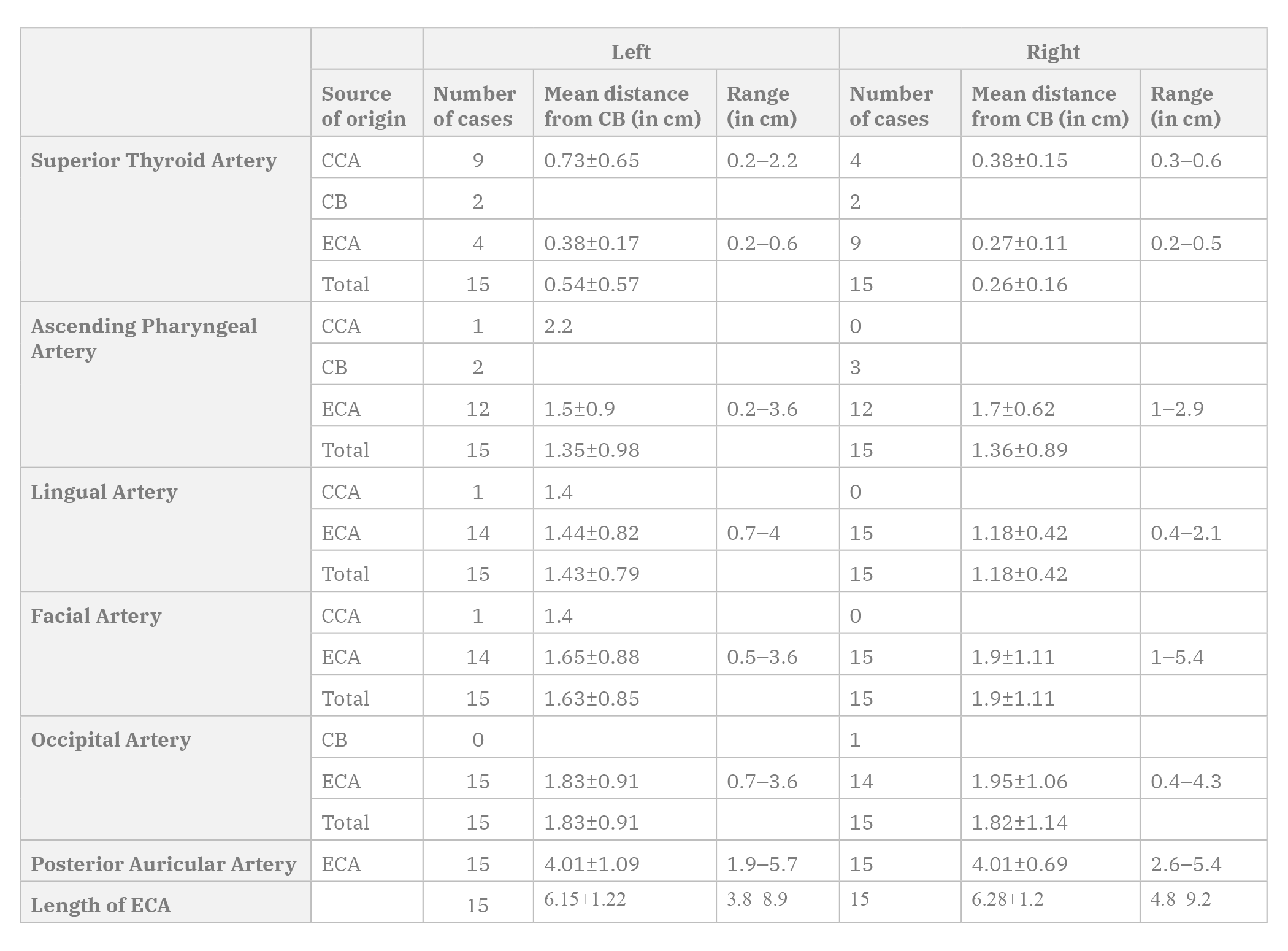
The ECA terminated as the maxillary artery and superficial temporal artery in all cases. The mean length of the ECA was 6.15±1.22 on the left and 6.28±1.2 on the right (Table 1). Table 1 shows the mean distance of origin of the arteries from the carotid bifurcation.
Common trunks of origin
In this study, there were several instances of branches of the ECA sharing common trunks of origin. Three types of common trunks were noted:
Linguo-facial trunk: between the lingual artery and the facial artery (Figs. 2B and 3A).
Pharyngo-occipital trunk: between the ascending pharyngeal artery and the occipital artery (Fig. 3B).
Auriculo-occipital trunk: between the posterior auricular artery and the occipital artery (Fig. 3A).
Linguo-facial trunk was the most common, present in 26.67% (8/30) cases at a mean distance of 1.3±0.31 cm from the carotid bifurcation. Five cases were seen on the left side, at a mean distance of 1.42±0.33 cm from the carotid bifurcation at a range of 0.9-1.8 cm. Three cases were seen on the right side, at a mean distance of 1.1±0.1 cm from the carotid bifurcation at a range of 1-1.2 cm. The linguo-facial trunk took origin from the ECA in all cases except one, where it took origin from the CCA.
Pharyngo-occipital trunk was seen in 23.33% (7/30) cases, at a mean distance of 2.23±0.97 cm from the carotid bifurcation. Four cases were seen on the left side, at a mean distance of 2±1.25 cm from the carotid bifurcation at a range of 0.7-3.6 cm. Three cases were seen on the right side, at a mean distance of 2.53±0.47 cm from the carotid bifurcation at a range of 2-2.9 cm. The pharyngo-occipital trunks took origin from the ECA in all cases.
Auriculo-occipital trunk was seen in 13.33% (4/30) cases at a mean distance of 3.48±0.57 cm from the carotid bifurcation. Two cases were seen on the left side, at a mean distance of 3.2±0.2 cm from the carotid bifurcation at a range of 3-3.4 cm. Two cases were seen on the right side, at a mean distance of 3.75±0.78 cm from the carotid bifurcation at a range of 3.2-4.3 cm. The auriculo-occipital trunk took origin from the ECA in all cases.
53.33% (16/30) cases showed the presence of at least one common trunk of origin. Presence of common trunks of origin was more common on the left side. These 16 cases included three, which showed the presence of more than one common trunk, two on the left and one on the right.
DISCUSSION
Surgeries like thyroidectomy, laryngectomy, faciomaxillary surgeries, tonsillectomy, glossectomy and other neck surgeries involve areas supplied by or related to branches of the ECA (Sanjeev et al., 2010). Preoperative appreciation of variations of the ECA can help in maintaining a bloodless field (Hansdak et al., 2015). Oro-facial reconstruction surgeries, including scalp transplantation, depend on the superior thyroid, lingual and facial arteries for their technical applicability, feasibility, and flap survival (Li et al., 2013; Siemionow and Kulahci, 2007).
Preoperative arteriograms are used to map out the vascularity and true extent of the head and neck tumors, while selective arterial embolization helps reduce their vascularity (Byun et al., 2012). Super-selective intra-arterial chemotherapy, aimed at treating head and neck tumors, like oral cancer, demands accurate knowledge of the arteries supplying the region (Tohnai, 2006).
Carotid bifurcation
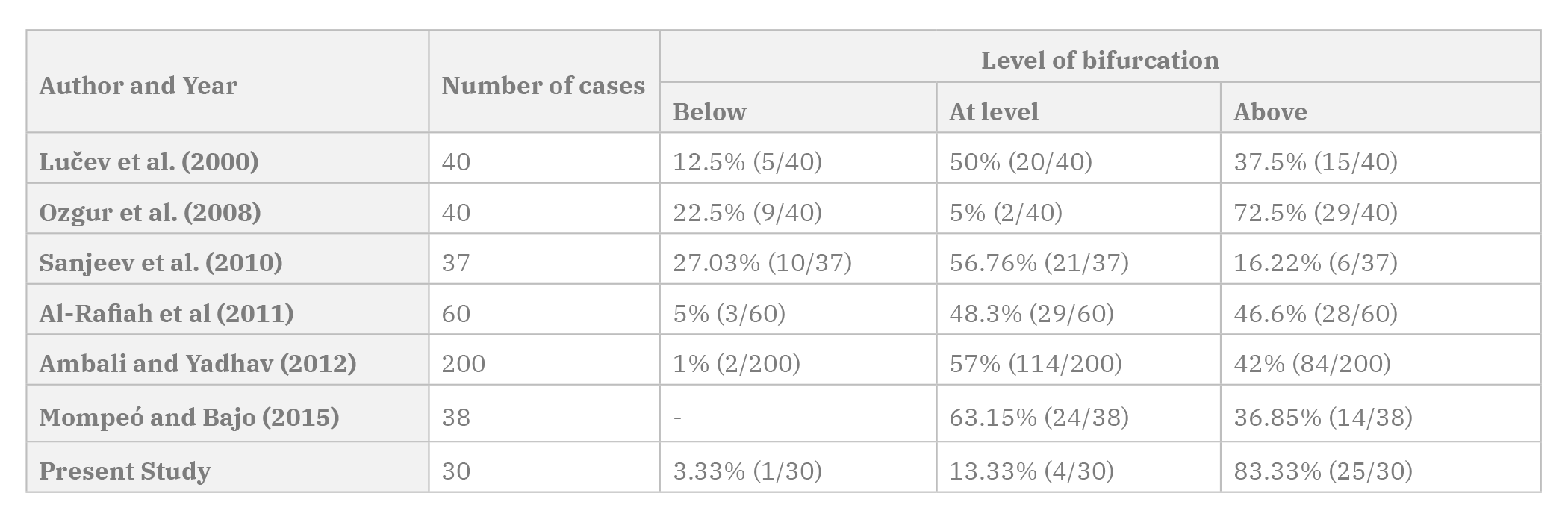
The CCA bifurcates into the ECA and ICA at the level of the superior border of the lamina of the thyroid cartilage. Previous literature shows that the level of carotid bifurcation is highly variable (Table 2). Cases of high bifurcation, low bifurcation, intrathoracic bifurcation and even the absence of CCA have been reported by various authors (Bergman et al., 1998; Cakirer et al., 2002; Chan et al., 2013; Gailloud et al., 2000; Gomez and Arnuk, 2013; Uzun et al., 2013). The present study showed a high number of high bifurcations, which were comparable to a study done by Ozgur et al. (2008a, b) in the Turkish population. In the present study, the distance of the bifurcation from the upper border of the thyroid cartilage was 1.22±1.03 cm on the left side, in comparison to 0.99±0.89 cm on the right side. According to Ribeiro et al. (2006), the carotid bifurcation levels were symmetrical on both sides.
Trifurcation of the CCA into ECA, ICA and a branch of ECA has been reported by various authors (Altinbas et al., 2015; Gürbüz et al., 2001). In the present study, trifurcation of CCA was seen in eight cases (26.67%). This included four cases of ascending pharyngeal artery, three cases of superior thyroid artery and one case of occipital artery taking origin from the carotid bifurcation. One case (3.33%) of tetrafurcation was seen in a female cadaver, where on the right side the superior thyroid artery and ascending pharyngeal artery took origin from the carotid bifurcation.
Superior thyroid artery
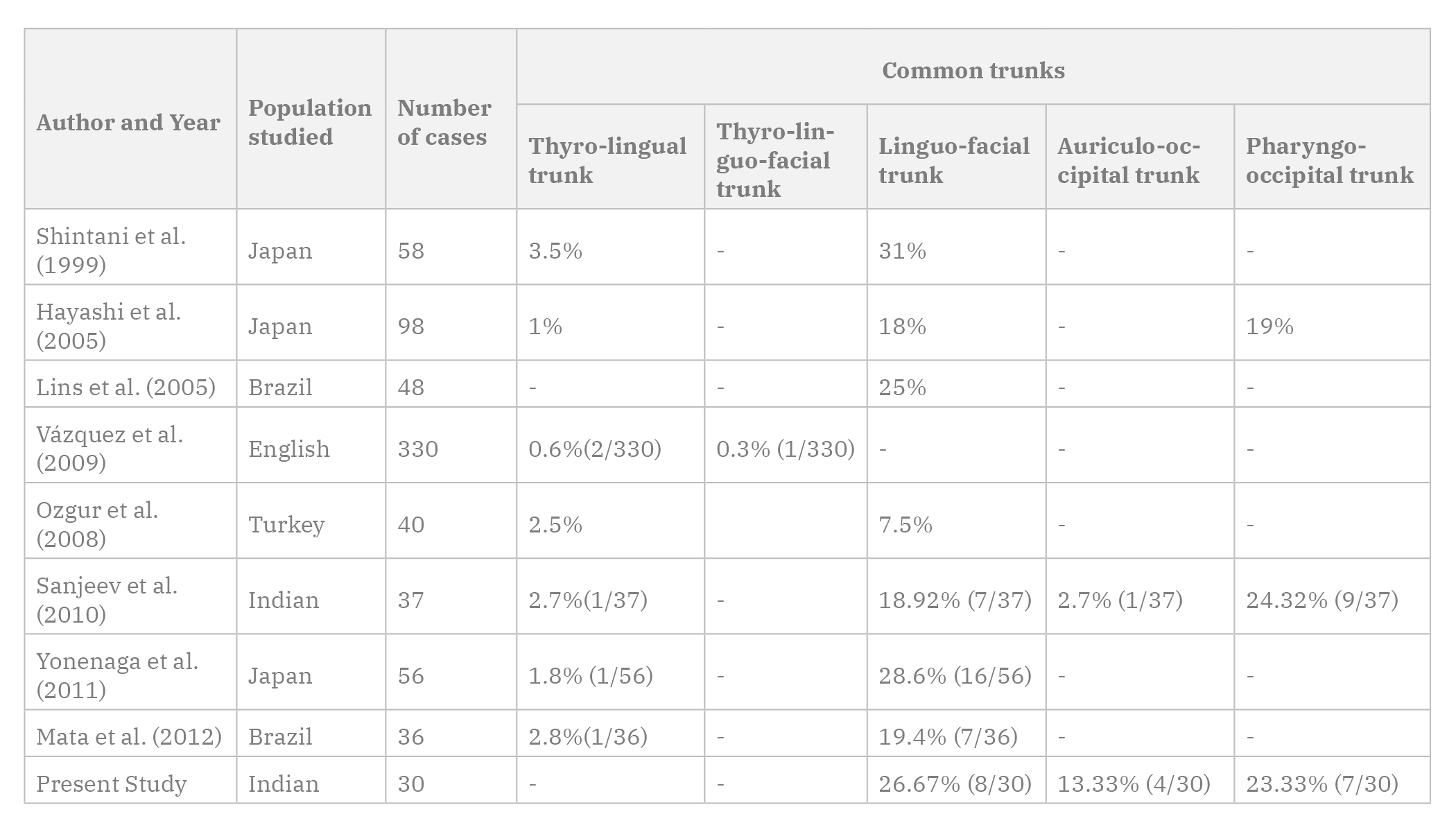
While most Indian authors have reported a low incidence of superior thyroid artery arising from the CCA, a higher incidence of 43.33% was seen in the present study done on the Punjabi population from India, which was similar to certain foreign authors (Table 3). This variation has been noted in classically referenced studies, with Quain (1844) reporting 14% cases, Livini (1903) reporting 9% cases and Adachi (1928) reporting 13% cases. The superior thyroid artery took origin from the CCA more commonly on the left side. The origin of the superior thyroid artery was at a mean distance of 0.4±0.43 cm from the carotid bifurcation, which was in accordance with Ozgur et al. (2009), who observed a similar result with a mean of 0.33±0.43 cm.
Ascending pharyngeal artery
In the present study, the ascending pharyngeal artery took origin from the ECA in 80% (24/30) cases. Variant origin from the carotid bifurcation was seen in 16.67% (5/30) cases and from CCA in 3.33% (1/30) cases (Fig. 2B).
The ascending pharyngeal artery took origin at a mean distance of 1.35±0.92 cm from the carotid bifurcation with a range of 0-3.6 cm, which was similar to Cavalcanti et al. (2009), who observed a similar result in the North American population with a mean of 1.53±0.83 cm with a range of 0-3.2 cm. The branches of the ascending pharyngeal artery anastomose with branches of ICA, maxillary artery, vertebral artery and occipital artery, thus providing important collateral circulation (Hacein-Bey et al., 2002).
Lingual artery
The lingual artery took origin from the ECA in all cases, except one, where it stemmed from the CCA along with the facial artery as a common trunk (Fig. 2B). Lučev et al. (2000) and Troupis et al. (2014), each observed a case where the lingual artery took origin from the carotid bifurcation, whereas Ambali and Jadhav (2012) observed a case in their study where the lingual artery took origin from the CCA. In the present study, the distance of origin of the lingual artery from the carotid bifurcation ranged from 0.4-4 cm, similar to the findings of Lins et al. (2005) and Lučev et al. (2000), where the range of origin was 0.5-3.7 cm and 0.5-4 cm respectively. The lingual artery was at a distance of 1.31±0.63 cm from the carotid bifurcation. Yonenaga et al. (2011) observed a mean distance of 2.13±0.85 cm in their study.
Facial artery
In the present study, the facial artery took origin from the CCA in one case (Fig. 2B). A Japanese study reported a mean distance of 3.08±1.18 cm from the carotid bifurcation, while in the present study, this distance was 1.77±0.98 cm (Yonenaga et al., 2011).
Occipital artery
In the present study, the occipital artery took origin from the ECA in all cases except one, where it took origin from the carotid bifurcation (Fig. 2A). Various authors have observed the occipital artery arising from the ICA (Adachi ,1928; Cappabianca et al., 2012; Small et al., 2014). Such a finding was absent in the present study. The origin of the occipital artery was at a mean distance of 1.83±1.01 cm from the carotid bifurcation with a range of 0-5.4 cm, indicating a wide range in the origin of occipital artery, whereas the observation made by Alvernia et al. (2006) showed a range of 0.7-2.8 cm with a mean of 1.74 cm.
Posterior auricular artery
In the present study the posterior auricular artery took origin from the ECA in all cases. In addition, the distance of origin of the posterior auricular arteries was similar on both sides.
Termination of external carotid artery
In the present study, all ECAs terminated as superficial temporal arteries and maxillary arteries. In one case observed by Sanjeev et al. (2010), the ECA trifurcated into the superficial temporal, maxillary and posterior auricular arteries. In the present study, the mean length of ECA was similar to the study by Sanjeev et al. (2010), who reported a mean length of 6 cm. A Japanese study by Yonenaga et al. (2011) showed the length to be 7.78±0.9 cm.
Common trunks of origin
Three varieties of common trunks were seen in the present study: linguo-facial trunks, pharyngo-occipital trunks and auriculo-occipital trunks.
As seen in Table 4, studies report a varying prevalence of branches of the ECA, originating through common trunks. This may depict a geographical variation in the branching pattern.
The vasculature of the head and neck is derived from two dorsal aortic arches along with two ventral pharyngeal arteries during early embryogenesis. These communicate through six aortic arches, which disappear variably during development. The carotid arterial system and its branches, with their variations, develop from this early system of arteries (Troupis et al., 2014).
Conclusions
In conclusion, differences in the branching pattern of the ECA are prevalent in the Indian Punjabi population. These include variations in the level of origin and common trunks. The typical level of origin at the upper border of the thyroid cartilage was seen only in four cases out of 30, with trifurcations seen in eight cases and tetrafurcation in one case. Linguo-facial trunk was seen in 26.67%, pharyngo-occipital trunk in 23.33% and auriculo-occipital trunk in 13.33% cases. Knowledge of these aberrations in the branching pattern of the ECA is crucial to clinicians and anticipating them will help in better diagnosis and treatment of patients.
Related articles
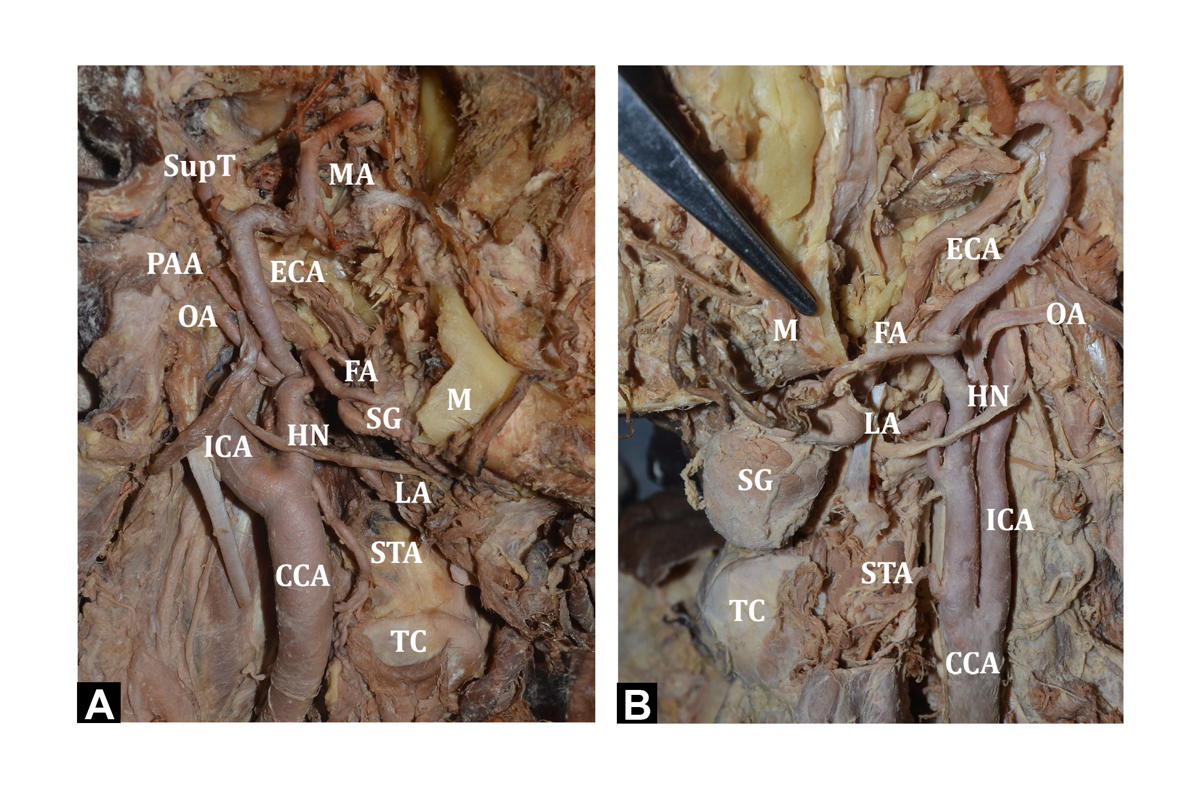 Fig. 1.- A) Showing the bifurcation of CCA above the level of TC (right side). B) Bifurcation of CCA below the upper border of TC (left side). TC - Thyroid cartilage; CCA - Common carotid artery; ECA - External carotid artery; ICA - Internal carotid artery; OA - Occipital artery; APA - Ascending pharyngeal artery; ComB - Communicating branch to ICA; BrICA - Branches arising from ICA; STA - Superior thyroid artery; LA - Lingual artery; FA - Facial artery; LFT – Linguo-facial trunk; AOT – Auriculo-occipital Trunk; PAA - Posterior auricular artery; SupT - Superficial temporal artery; MA - Maxillary artery; POT - Pharyngo-occipital trunk HN – Hypoglossal nerve; SG – Submandibular gland.
Fig. 1.- A) Showing the bifurcation of CCA above the level of TC (right side). B) Bifurcation of CCA below the upper border of TC (left side). TC - Thyroid cartilage; CCA - Common carotid artery; ECA - External carotid artery; ICA - Internal carotid artery; OA - Occipital artery; APA - Ascending pharyngeal artery; ComB - Communicating branch to ICA; BrICA - Branches arising from ICA; STA - Superior thyroid artery; LA - Lingual artery; FA - Facial artery; LFT – Linguo-facial trunk; AOT – Auriculo-occipital Trunk; PAA - Posterior auricular artery; SupT - Superficial temporal artery; MA - Maxillary artery; POT - Pharyngo-occipital trunk HN – Hypoglossal nerve; SG – Submandibular gland.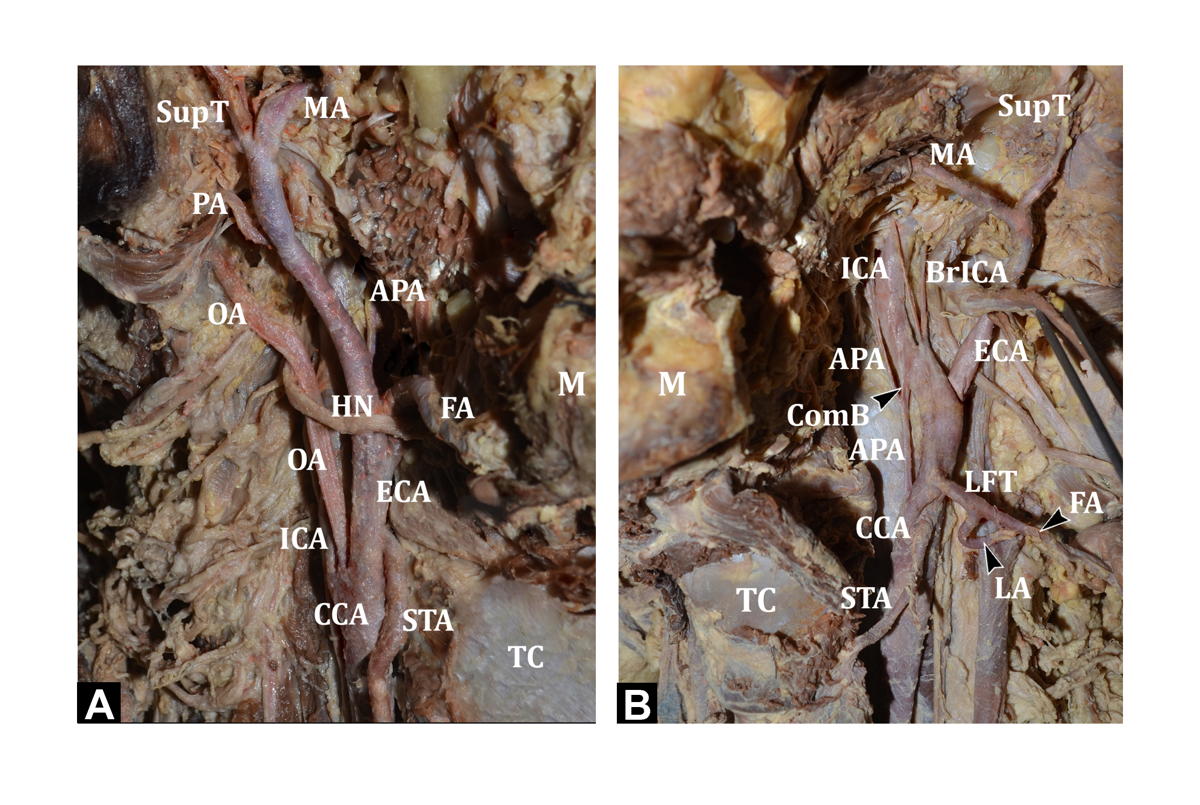 Fig. 2.- A) Showing the OA arising at the level of carotid bifurcation (right side). B) Showing APA arising from CCA with a communicating branch to ICA. In addition, LFT is seen arising from the CCA. ICA gives two cervical branches (left side).TC - Thyroid cartilage; CCA - Common carotid artery; ECA - External carotid artery; ICA - Internal carotid artery; OA - Occipital artery; APA - Ascending pharyngeal artery; ComB - Communicating branch to ICA; BrICA - Branches arising from ICA; STA - Superior thyroid artery; LA - Lingual artery; FA - Facial artery; LFT – Linguo-facial trunk; AOT – Auriculo-occipital Trunk; PAA - Posterior auricular artery; SupT - Superficial temporal artery; MA - Maxillary artery; POT - Pharyngo-occipital trunk HN – Hypoglossal nerve; SG – Submandibular gland.
Fig. 2.- A) Showing the OA arising at the level of carotid bifurcation (right side). B) Showing APA arising from CCA with a communicating branch to ICA. In addition, LFT is seen arising from the CCA. ICA gives two cervical branches (left side).TC - Thyroid cartilage; CCA - Common carotid artery; ECA - External carotid artery; ICA - Internal carotid artery; OA - Occipital artery; APA - Ascending pharyngeal artery; ComB - Communicating branch to ICA; BrICA - Branches arising from ICA; STA - Superior thyroid artery; LA - Lingual artery; FA - Facial artery; LFT – Linguo-facial trunk; AOT – Auriculo-occipital Trunk; PAA - Posterior auricular artery; SupT - Superficial temporal artery; MA - Maxillary artery; POT - Pharyngo-occipital trunk HN – Hypoglossal nerve; SG – Submandibular gland. Fig. 3.- A) Showing the LA and FA arising from the LFT and OA and PAA arising from AOT (right side). B) Showing the APA and OA arising from the POT (left side). TC - Thyroid cartilage; CCA - Common carotid artery; ECA - External carotid artery; ICA - Internal carotid artery; OA - Occipital artery; APA - Ascending pharyngeal artery; ComB - Communicating branch to ICA; BrICA - Branches arising from ICA; STA - Superior thyroid artery; LA - Lingual artery; FA - Facial artery; LFT – Linguo-facial trunk; AOT – Auriculo-occipital Trunk; PAA - Posterior auricular artery; SupT - Superficial temporal artery; MA - Maxillary artery; POT - Pharyngo-occipital trunk HN – Hypoglossal nerve; SG – Submandibular gland.
Fig. 3.- A) Showing the LA and FA arising from the LFT and OA and PAA arising from AOT (right side). B) Showing the APA and OA arising from the POT (left side). TC - Thyroid cartilage; CCA - Common carotid artery; ECA - External carotid artery; ICA - Internal carotid artery; OA - Occipital artery; APA - Ascending pharyngeal artery; ComB - Communicating branch to ICA; BrICA - Branches arising from ICA; STA - Superior thyroid artery; LA - Lingual artery; FA - Facial artery; LFT – Linguo-facial trunk; AOT – Auriculo-occipital Trunk; PAA - Posterior auricular artery; SupT - Superficial temporal artery; MA - Maxillary artery; POT - Pharyngo-occipital trunk HN – Hypoglossal nerve; SG – Submandibular gland.ACAR M, SALBACAK A, SAKARYA ME, ZARARSIZ I, ULUSOY M (2013) The morphometrical analysis of the external carotid artery and its branches with multidetector computerized tomography angiography technique. Int J Morphol, 31: 1407-1414.
ADACHI B (1928) Das arteriensystem der Japaner. Maruzen, Kyoto, pp 54-82.
AL-RAFIAH A, EL-HAGGAGY AA, AAL IHA, ZAKI AI (2011) Anatomical study of the carotid bifurcation and origin variations of the ascending pharyngeal and superior thyroid arteries. Folia Morphol, 70: 47-55.
ALTINBAS NK, UNAL S, PEKER A, UZUN C, AKKAYA Z, PEKER E (2015) Trifurcation of the left common carotid artery. Pol J Radiol, 80: 376-378.
ALVERNIA JE, FRASER K, LANZINO G (2006) The occipital artery: a microanatomical study. Oper Neurosurg, 58: 114-122.
AMBALI M, JADHAV SJ (2012) Variations in bifurcation point and branching pattern of common carotid arteries: a cadaveric study. J Pharm Biomed Sci, 25: 147-151.
BERGMAN RA, THOMPSON SA, AFIFI AK, SAADEH (1998) Compendium of human anatomic variation. Urban & Schwarzenberg, Baltimore-Munich, pp 28-31.
BYUN H, CHO YS, CHU HS (2012) Glomus faciale: primary paraganglioma of the facial canal. J Int Adv Otol, 8: 137-142.
CAKIRER S, KARAARSLAN E, KAYABALI M, ROZANES I (2002) Separate origins of the left internal and external carotid arteries from the aortic arch: MR angiographic findings. Am J Neuroradiol, 23: 1600-1602.
CAPPABIANCA S, SCUOTTO A, IASELLI F, SPINAZZOLA NPD, URRARO F, SARTI G, MONTEMARANO M, GRASSI R, ROTONDO A (2012) Computed tomography and magnetic resonance angiography in the evaluation of aberrant origin of the external carotid artery branches. Surg Radiol Anat, 34: 393-399.
CAVALCANTI DD, REIS CVC, HANEL R, SAFAVI-ABBASI S, DESHMUKH P, SPETZLER RF, PREUL MC (2009) The ascending pharyngeal artery and its relevance for neurosurgical and endovascular procedures: Oper Neurosurg, 65 ONS Suppl 1: ons114-ons120.
CHAN YC, WONG WH, CHENG SW (2013) Successful carotid endarterectomy in a patient with an aberrant branch from the common carotid artery. Ann R Coll Surg Engl, 95: e17-e19.
GAILLOUD P, MURPHY KJ, RIGAMONTI D (2000) Bilateral thoracic bifurcation of the common carotid artery associated with Klippel-Feil anomaly. Am J Neuroradiol, 21: 941-944.
GERMANS MR, REGLI L (2014) Posterior auricular artery as an alternative donor vessel for extracranial-intracranial bypass surgery. Acta Neurochir (Wien), 156: 2095-2101.
GOMEZ CK, ARNUK OJ (2013) Intrathoracic bifurcation of the right common carotid artery. BMJ Case Rep, 2013: bcr2012007554.
GÜRBÜZ J, CAVDAR S, OZDOĞMUŞ O (2001). Trifurcation of the left common carotid artery: a case report. Clin Anat, 14: 58-61.
HACEIN-BEY L, DANIELS DL, ULMER JL, MARK LP, SMITH MM, STROTTMANN JM, BROWN D, MEYER GA, WACKYM PA (2002) The ascending pharyngeal artery: branches, anastomoses, and clinical significance. Am J Neuroradiol, 23: 1246-1256.
HANSDAK R, RUSTAGI SM, LOH HK, PAKHIDDEY R, SURI RK, MEHTA V (2015) Variant topography of the carotid arteries - a clinicoanatomical appraisal. Int J Adv Res, 3: 1-6.
HAYASHI N, HORI E, OHTANI Y, OHTANI O, KUWAYAMA N, ENDO S (2005) Surgical anatomy of the cervical carotid artery for carotid endarterectomy. Neurol Med Chir (Tokyo), 45: 25-30.
LINS CCDSA, CAVALCANTI JS, NASCIMENTO DLD (2005) Extraoral ligature of lingual artery: anatomic and topographic study. Int J Morphol, 23: 271-274.
LIVINI F. (1903) Le type normal et les variations de l’A. carotis externa. Arch Ital Biol, 39: 486-487.
LO A, OEHLEY M, BARTLETT A, ADAMS D, BLYTH P, AL-ALI S (2006) Anatomical variations of the common carotid artery bifurcation. ANZ J Surg, 76: 970-972.
LUČEV N, BOBINAC D, MARIĆ I, DREŠĆIK I (2000) Variations of the great arteries in the carotid triangle. Otolaryngol Head Neck Surg, 122: 590-591.
MATA JR, MATA FR, SOUZA MCR, NISHIJO H, FERREIRA TAA (2012) Arrangement and prevalence of branches in the external carotid artery in humans. Ital J Anat Embryol, 117: 65-74.
MOMPEÓ B, BAJO E (2015) Carotid bifurcation - clinical relevance. Eur J Anat, 19: 37-45.
OZGUR Z, GOVSA F, OZGUR T (2008a) Anatomic evaluation of the carotid artery bifurcation in cadavers: implications for open and endovascular therapy. Surg Radiol Anat, 30: 475-480.
OZGUR Z, GOVSA F, OZGUR T (2008b) Assessment of origin characteristics of the front branches of the external carotid artery. J Craniofac Surg, 19: 1159-1166.
OZGUR Z, GOVSA F, CELIK S, OZGUR T (2009) Clinically relevant variations of the superior thyroid artery: an anatomic guide for surgical neck dissection. Surg Radiol Anat, 31: 151-159.
PATEL JP, DAVE RV, SHAH RK, KANANI SD, NIRVAN AB (2013) A study of superior thyroid artery in 50 cadavers. Int J Biol Med Res, 4: 2875-2878.
QUAIN R (1844) The anatomy of the arteries of the human body. Taylon and Walton, London, pp 55-112.
RAJAMANI K, CHATURVEDI S (2011) Stroke prevention-surgical and interventional approaches to carotid stenosis. Neurother J Am Soc Exp Neurother, 8: 503-514.
RIBEIRO RA, RIBEIRO JADS, FILHO OAR, CAETANO AG, FAZAN VPS (2006) Common carotid artery bifurcation levels related to clinical relevant anatomical landmarks. Int J Morphol, 24: 413-416.
SANJEEV IK, ANITA H, ASHWINI M, MAHESH U, RAIRAM GB (2010) Branching pattern of external carotid artery in human cadavers. J Clin Diag Res, 4: 3128-3133.
SHINTANI S, TERAKADO N, ALCALDE RE, TOMIZAWA K, NAKAYAMA S, UEYAMA Y, ICHIKAWA H, SUGIMOTO T, MATSUMURA T (1999) An anatomical study of the arteries for intraarterial chemotherapy of head and neck cancer. Int J Clin Oncol, 4: 327-330.
SHIVALEELA C, ANUPAMA D, LAKSHMI PSR (2016) Study of anatomical variations in the origin of superior thyroid artery. Int J Anat Res, 4: 1765-1768.
SIEMIONOW M, KULAHCI Y (2007) Facial transplantation. Semin Plast Surg, 21: 259-268.
SMALL JE, HARRINGTON J, WATKINS E (2014) Prevalence of arterial branches arising from the extracranial internal carotid artery on CT angiography. Surg Radiol Anat, 36: 789-793.
TOHNAI I (2006) Chemotherapy using intra-arterial infusion for oral cancer. Nagoya J Med Sci, 68: 101-108.
TROUPIS T, MICHALINOS A, DIMOVELIS I, DEMESTICHA T, VLASIS K, SKANDALAKIS P (2014) Bilateral abnormal origin of the anterior branches of the external carotid artery. Ann Vasc Surg, 28: 494.e5-494.e7.
UZUN L, KOKTEN N, KILICASLAN A, TASEL B, KALCIOGLU MT, TEKIN M (2013) Bilateral lower cervical bifurcation of the common carotid artery. Case Rep Otolaryngol, 2013: 1-3.
VÁZQUEZ T, COBIELLA R, MARANILLO E, VALDERRAMA FJ, MCHANWELL S, PARKIN I, SAÑUDO JR (2009) Anatomical variations of the superior thyroid and superior laryngeal arteries. Head Neck, 31: 1078-1085.
WON SY (2016) Anatomical considerations of the superior thyroid artery: its origins, variations, and position relative to the hyoid bone and thyroid cartilage. Anat Cell Biol, 49: 138-141.
YONENAGA K, TOHNAI I, MITSUDO K, MORI Y, SAIJO H, IWAI T, YONEHARA Y, OTA Y, TORIGOE K, TAKATO T (2011) Anatomical study of the external carotid artery and its branches for administration of superselective intra-arterial chemotherapy via the superficial temporal artery. Int J Clin Oncol, 16: 654-659.