The pterion and asterion are at the outer surface of the skull. The pterion is the region where the frontal, sphenoid, parietal and temporal bones join together, and the asterion is the intersection of parietal, temporal and occipital bones. The sutural pattern of both are different in the various population and races. This study examines the patterns of suture of the pterion and asterion in 210 semi-skulls of 146 males and 64 females cadaver. Four main types of anatomical patterns were found: sphenoparital 84.2%, frontotemporal 8.1%, star shaped 3.5% and epipteric 2.4%. In the study of asterion, in 14.7%, type I, and 86.3% type II was observed. In the first study of the sutural pattern of the pterion and asterion in Iranian population, similarity to the other studies of the Indian and Middle Eastern regions was observed.
The morphology of the pterion and asterion sutures in Iranian population
Hossein Jafari Marandi 1, Jaber Gharehdaghi 2, Forouzan Fars 3, Masoud Zeinali 1, Hosein Safari 1
1 Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran
2 Iran Legal Medicine Organization, Tehran, Iran
3 Iran University of Medical Sciences, Tehran, Iran
SUMMARY
Sign up or Login
INTRODUCTION
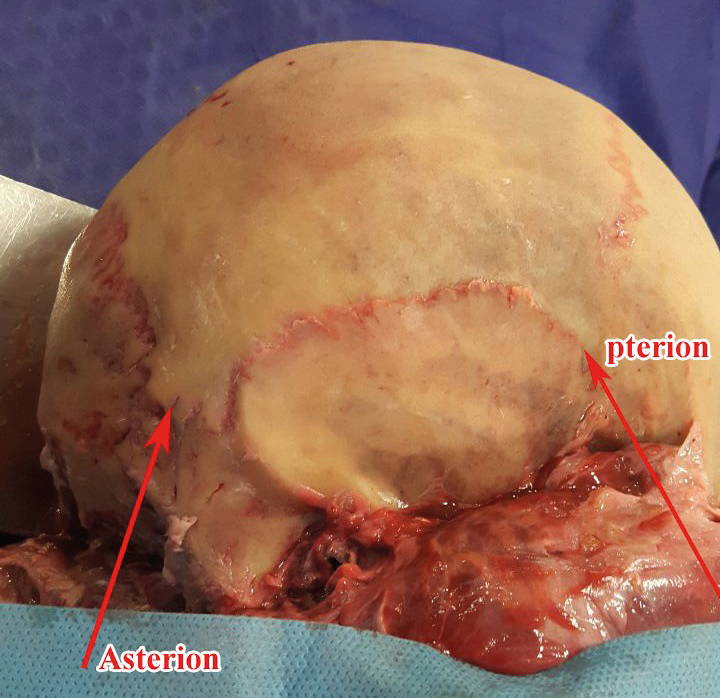
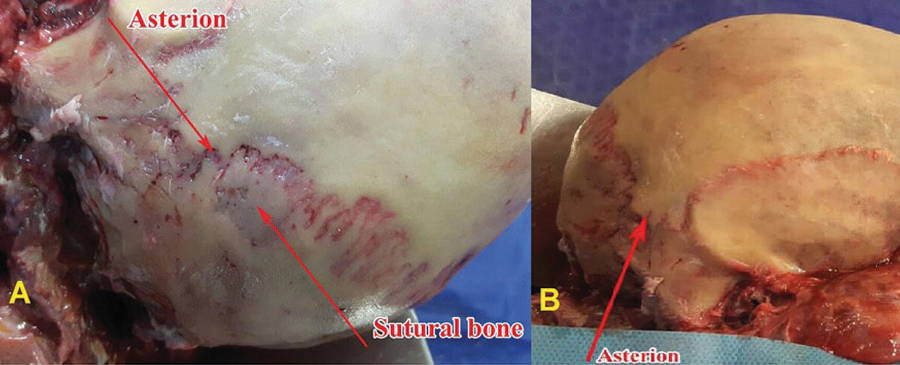
The pterion is adjacent to the temporal cavity and the binding site of the frontal, parietal, temporal, and sphenoid bone, and is located in the anterolateral part of the skull, which is differing among primates. In the asterion, the connection between the parietal, temporal and occipital bones is created (Fig. 1). The variation in the pattern of the pterion has a surgical importance to deep-seated structures. The stability of sutures in animals in a particular species raises the genetic basis (Williams et al., 1998).
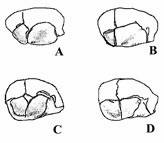
In 1956, Murphy and coworkers described four types of pterion (Murphy, 1956). Based on this classification, the sphenoid bone is connected to the parietal bone, and the suture is of sphenoparital type, and this prevents the attachment of frontal and parietal bones to each other. In the type of fronto-temporal, frontal and temporal bones contact each other and prevent the attachment of frontal and parietal bones (Kumar and Mahajan, 2014). In the star-shaped type, the irregular shape of the sutures are similar to the letter K, and the bones meet at one point. It should be noted that in the area of the pterion region there is also the possibility of developing a sutural bone (epipteric type) (Kumar and Mahajan, 2014) (Fig. 2).
Occasionally, bone formation may also occur in the vicinity of the septum of another bony building (sutural bone). Although the authors believe that the sutural bone is a natural and gene-controlled process, studies also emphasize the role of pathologies such as hydrocephalus, as well as studies focusing on the role of ethnic and racial differences in the incidence of sutures forms (Havaldar et al., 2015). The shape of the asterion and pterion sutures are important in the neurosurgical approaches to the cranial cavity (Ersoy et al., 2003; Ucerler and Govsa, 2006; Galindo de León et al., 2013; Havaldar et al., 2015; Natekar et al., 2018).The current study examines the forms of sutures of the pterion and asterion point in the Iranian population.
MATERIALS AND METHODS
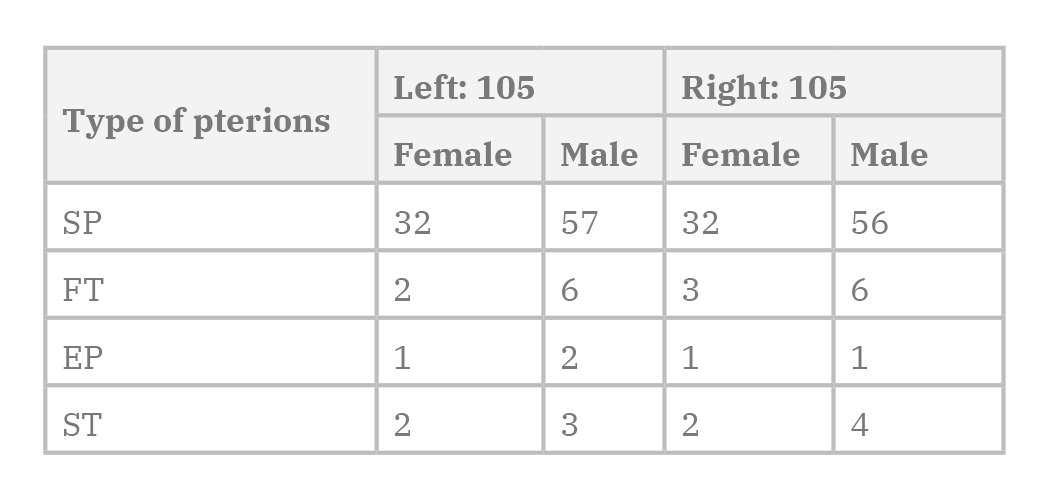
After obtaining the necessary authorization, 210 adult semi-skulls, including 69.5% males and 30.5% females, were examined. In this study, asterion sutures were classified into 2 types: type I which had a sutural bone and type II without sutural bone (Fig. 3).
Each suture point of the pterion was divided into sphenoparital, fronto-temporal, stellar and epipteric (sutural). In each sample, soft tissue was removed from the suture points using appropriate tools and then the sutures were examined. In this research, descriptive statistics (frequency, mean, variance, standard deviation) were used to analyze the data. To analyze the demographic characteristics of the research, statistical analysis with SPSS software was used.
RESULTS
Asterion
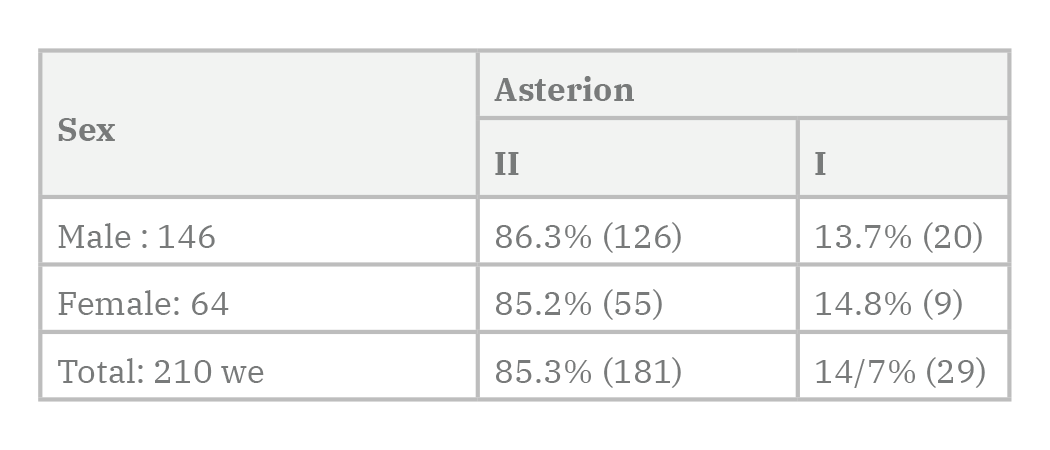
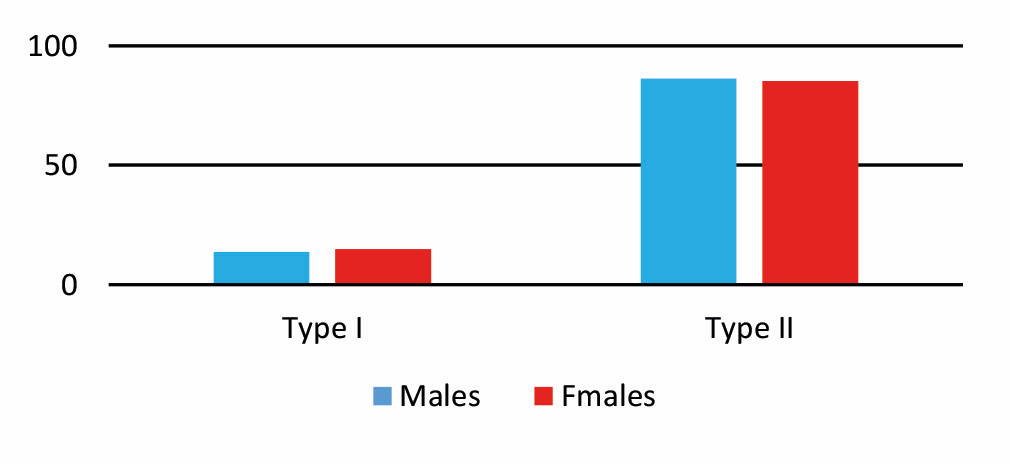
Regarding the results of the study, 210 semi-skull specimens were categorized in 181 specimens (85.3% of type II) and 29 specimens (14.7% of type I) asterions.
Among the females, type I of asterion (14.8%) was slightly more common than in men (13.7%). However, there was no significant difference between the two sexes. Among the tested specimens, asterion type II was more common in men with 86.3% than women with 85.2%. However, there was no significant difference between the two sexes (Tables 1-2 and 5, and Diagrams 1-2).
Pterion
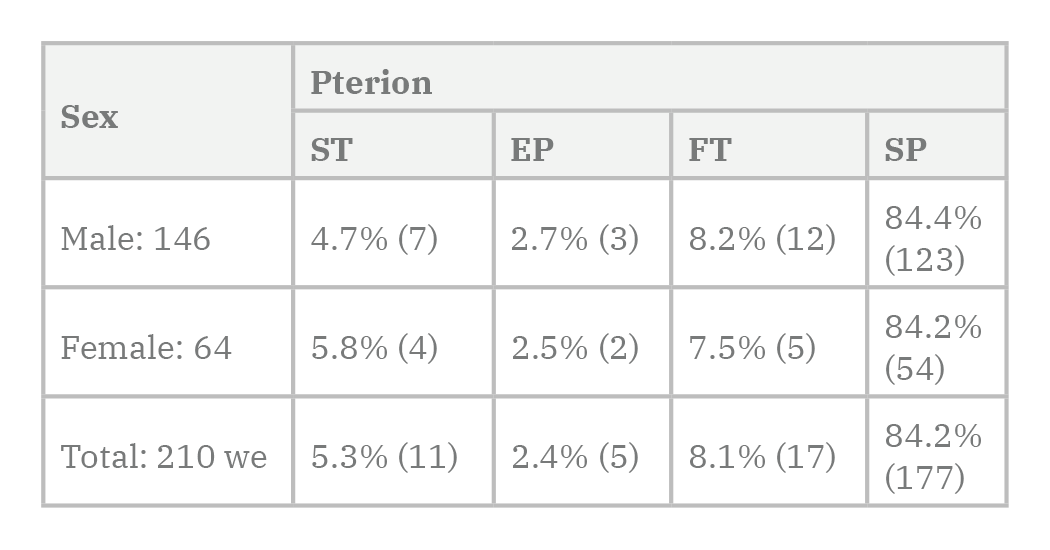
According to the results, based on the 210 samples studied, 177 specimens (84.2%) were found to have sphenopariatal, of which 84.4% were male and 84.2% were female respectively. This type was the most common form of the pterion in this study. Also, the epipteric type of the pterion with 2.4% (five samples) was the lowest form of pterion in the studied samples.
Sexual difference in pterion types was not observed between the studied samples, although the star-shaped type of pterion in females was 5.8%, more than that of the male, 4.7% (Tables 3-4 and 6 and Diagrams 3-4).
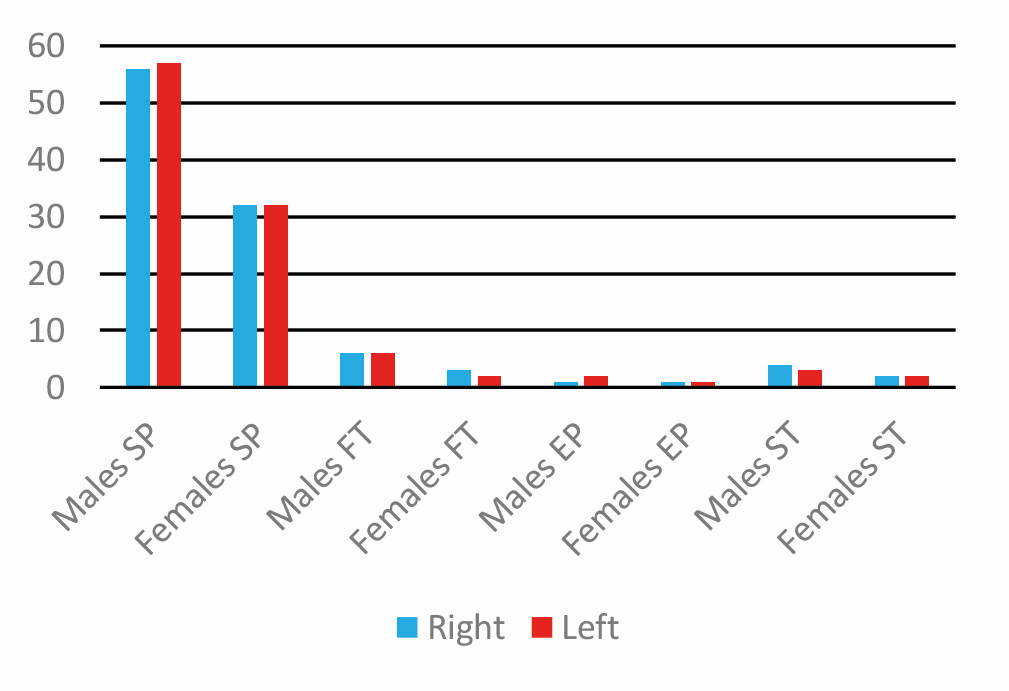
According to the results of 210 semi-skulls of pterions in the right and left hemispheres, the frontotemporal type in the right semi-skull hemisphere was more common in females than in males. Also, the star-shaped pterion in the right hemisphere of males is more common than in females. However, there is no significant difference between the two types of sex and semi-skull of epipteric and star shaped of pterions.
DISCUSSION
The findings of this study are in line with previous studies, although some differences are evident. Ashley’s study on the subject of pterion from an anthropological perspective points to the higher percentage of epipteric sutures in the human societies (Ashley-Montagu, 1933). In this study, the findings were in the period between 1772 and 1930, which contains important points in the pattern of the pterion in humans. He identified sixteen general patterns of pterion.
Murphy (1956) studied four main patterns of pterion and five basic patterns for epipteric sutures. Wang et al. (2006), studying the skull sutures between the monkeys and observing the pattern of family gathering, suggested that this diversity was inherited. Liu et al. (1995), in a study on mice, emphasizes the role of temporal rotation in the development of sutures and the adaptation of skull growth.
Liu et al. (1999), showed that increased activity of the MSX2 gene plays a direct role in the morphogenesis of skull bones. In other words, according to the results of morphological and genetic studies, the pattern of suture formation is influenced by ethnic and environmental factors.
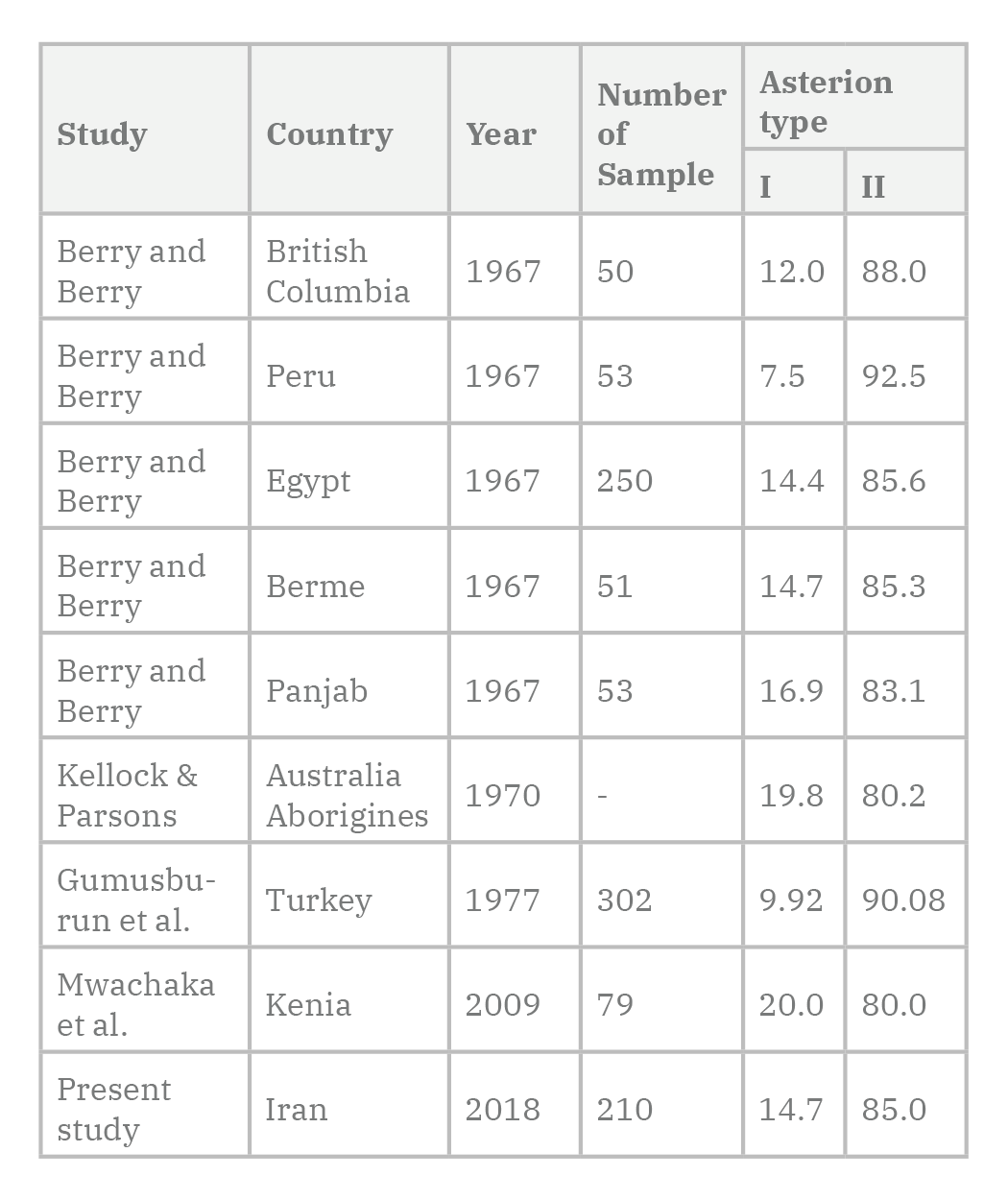
In the current study, the prevalence of type I asterion was 14.7%, which is equivalent with the study by Berry and Berry (1967) in Egyptian and Indian populations. However, in the study of Mwachaka et al. (2009), asterion type 1 was reported in 20% of cases. In the recent study, the sample of asterion type 1 was lower in women, with no difference in men on the right and left side of the skull. Singh (2012), in a study on 55 skull cases, reported an asterion type I occurrence of 16.36% in samples. In this study, type I asterion in the left side was lower in men than in women.
Havaldar et al. (2015), in a study on the morphology of asterion sutures in India, which were performed on 250 skulls, asterion type I was seen in 18% of men and 20% of women, and also the type II in 82% of men and 80% of women was seen. There was no difference in the asterion morphology in the Indian population compared to other populations. In the study of Galindo-de León et al. (2013), asterion type I was seen in 74.4% of the cases.
The study of Deepak and Dakshayani (2015), which was designed on 50 skulls of both genders to assess the type of asterion, and the importance of bony landmarks in relation to adjacent venous sinus structures, type II (lack of sutural bone) with 73% was more common than type I. In the present study, 85.3% of the cases were asterion type II specimens. This review is consistent with previous studies.
In the Murphy (1956) study, which was conducted among native Australians, 75% of the specimens were sphenoparietal sutures. Saxena et al. (1988) and Asala and Mbajiorgu (1996), in the Nigerian, Southern and Northern Indian populations, reported prevalence of sphenoparietal suture between 82.1 and 93.5%, respectively. In studies by Zalawadia et al. (2010), Mwachaka et al. (2009), Hussain et al. (2011) and Ukoha et al. (2013) that were conducted in populations in Kenya, India and Nigeria, the sphenoparietal sutures values was reported between 66 to 91.7%.
The studies of Ersoy et al. (2003) and Ogouz et al. (2004), which were performed on the Turkish population, reported sphenoparietal sutures values between 87.35% and 88%. The highest levels of sphenoparietal sutures were reported by Saxena et al. (1988) with 95.3% in the Indian population, and the lowest in 66% of cases in the study of Mwachacka et al. (2009) in Kenya. According to the results of this study, we can say that sphenoparietal suture has been observed in 84.2% of cases, which is in accordance with previous studies.
Ersoy et al. (2003) performed a study on 490 adult mature skulls. In this study, 9% of cases (44 out of 490 cases) were of epipteric bone. They concluded that there is a higher possibility of unwanted entry into the orbital cavity in epipteric skulls with anterior position. The highest rates of epipteric sutures reported by Lee et al. (2001) that was 40.3%. In the current study, an amount of 2.4% was obtained for epipteric sutures.
In the study of fronto-temporal sutures, the highest prevalence (15%) was reported in the study by Mwachaka et al. (2009) and in Lee et al. (2001) studying the Korean population, in which no fronto-temporal sutures were seen. In this study, the incidence of fronto-temporal sutures was found to be 8.1% among the studied population.
In the study of the Matsumura et al. (1991), the star-shaped sutures, 17.7%, reported the highest prevalence rates among the populations. In the studies of Asala and Mbajiorgu (1996), and Lee et al. (2001), on Nigerian and Korean populations, the star-shaped sutures were not seen.
In the study of epipteric sutures, a large dispersion has been reported. In Lee et al. (2001), epipteric sutures were reported as 40.3%, which was the highest percentage reported among different populations. Also, in the studies of Saxena et al. (2003) and Oguz et al. (2004), this gap was not seen in the population of South India and Turkey. In the current study, epipteric suture was observed with 2.4% in the studied population.
Conclusion
The shape of the pterion and asterion sutures in the Iranian population is in line with other populations. Since both major sutures are under investigation, important landmarks are in the surgical approach. The prevalence of sutural bones (type I in asterion and the epipteric in the pterion) may affect the surgical approach.
Related articles
ASALA SA, MBAJIORGU FE (1996) Epigenetic variations in the Nigerian skull: sutural pattern at the pterion. East African Med J, 73(7): 484-486.
ASHLEY-MONTAGU MF (1933) The anthropological significance of the pterion in the primates. Am J Phys Anthropol, Vol. XVIII, no. 2.
BERRY CA, BERRY RJ (1967) Epigenetic variation in the human cranium. J Anat, 101(2): 361-379.
CHARULATA S, ABHILASHA W (2015) To study the morphology of pterion in dry human skull in Vidarbha Region. Int J Sci Res (IJSR), 4(1).
DEEPAK S, DAKSHAYANI KR (2015) Morphometric features of asterion in adult human skulls. Int J Res Med Sci, 3(6): 1325-1328.
ERSOY M, EVLIYAOGLU C, BOZKURT MC, KONUKSAN B, TEKDEMIR I, KESKIL IS (2003) Epipteric bones in the pterion may be surgical pitfall. Minim Invasive Neurosurg, 46: 364-365.
GALINDO-DE LEÓN S, HERNÁNDEZ-RODRÍGUEZ AN, MORALES-ÁVALOS R, THERIOT-GIRÓN MC, ELIZONDO-OMAÑA RE, GUZMÁN-LÓPEZ S (2013) Morphometric characteristics of the asterion and the posterolateral surface of the skull: relationship with dural venous sinuses and neurosurgical importance. Cir Cir, 81: 269-273.
GUMUSBURUN E, SEVIM A, KATKICI U, ADIGUZEL E, GULEC E (1997) A study of sutural bones in Anatolian-Ottoman skulls. Int J Anthropol, 12(2): 43-48.
HAVALDAR PP, SHRUTHI BN, SAHEB SH, HENJARAPPA KS (2015) Morphological study on types of asterion. Int J Intg Med Sci, 2(10): 167-69.
HUSSAIN SS, MAVISHETTER GF, THOMAS ST, PRASANNA LC, MURALIDHAR P (2011) A study of sutural morphology of the pterion and asterion among human Indian skulls. Biomed Res, 22: 73-75.
KELLOCK WL, PARSONS PA (1970) A comparison of the incidenceof minor nonmetrical cranial variants in Australian Aborigines with those of Melanesia and Polynesia. Am J Phys Anthropol, 33(2): 235-239.
KUMAR S, MAHAJAN A (2014) Pterion formation in North Indian Population: An anatomico-clinical study. Int J Morphol, 32(4): 1444.
LEE UY, PARK DK, KWON SO, PAIK DJ, HAN SH (2001) Morphological analysis of the pterion in Korean. Korean J Phys Anthropol, 14: 281-289.
LIU YH, KUNDU R, WU L, LUO W, IGNELZI JR MA, SNEAD ML, MAXSON JR ME (1995) Premature suture closure and ectopic cranial bone in mice expressing Msx2 transgenes in the developing skull. Proc Natl Acad Sci USA, 92: 6137-6141.
LIU YH, TANG Z, KUNDU RK, WU L, LUO W, ZHU D, SANGIORGI F, SNEAD ML, MAXSON RE (1999) Msx2 gene dosage influences the number of proliferative osteogenic cells in growth centers of the developing murine skull: a possible mechanism for MSX2-mediated craniosynostosis in humans. Develop Biol, 205: 260-274.
MANJUNATH KY, THOMAS IM (1993) Pterion variants and epiptericossicles in South Indian skulls. J Anat Soc India, 42: 85-94.
MATSUMARA G, KIDA K, ICHIKAWA R, KODAMA G (1991) Pterion and epipteric bones in Japanese adults and fetuses with special reference to their reference to their formation and variations. Acta Anat Nippon, 66: 462-471.
MURPHY T (1956) The pterion in Australian Aborigine. Am J Phys Anthropol, 14(2): 225-244.
MWACHAKA PM, HASSANALI J, ODULA P (2009) Sutural morphology of the pterion and asterion among adult Kenyans. Braz J Morphol Sci, 26(1): 4-7.
NATEKAR PE, DESOUZA FM, NATEKAR SP (2018) Pterion: An anatomical variation and surgical landmark. www: indianjotol.org (Accesed on April 26) IP: 178.131.81.57.
OGUZ O, SANLI SG, BOZKIR MG, SOAMES RW (2004) The pterion in Turkish male skulls. Surg Radiol Anat, 26: 220-224.
SAXENA SK, JAIN SP, CHOWDHARY DS (1988) A comparative study of pterion formation and its variations in the skulls of Nigerians and Indians. Anthropol Anz, 46: 75-82.
SAXENA RC, BILODI AKS, MANE SS, KUMAR A (2003) Study of pterion in skulls of awadh area-in and around Lucknow. Kathmandu Univ Med J, 1: 32-33.
SINGH R (2012) Incidence of sutural bones at asterion in adults indian skulls. Int J Morphol, 30(3): 1182-1186.
SUDHA R, SRIDEVI C, EZHILARASI M (2013) Anatomical variation in the formation of pterion and asterion in south Indian population. Int J Curr Res Rev, 5(9): 93-98.
UCERLER H, GOVSA F (2006) Asterion as a surgical landmark for lateral cranial base approaches. J Cranio-Maxillofac Surg 34: 415-420.
UKOHA U, ORANUSI CK, OKAFOR JI, UDEMEZUE OO, ANYABOLU AE, NWAMARACHI TC (2013) Anatomic study of the pterion in Nigerian dry human skulls. Nigerian J Clin Pract, 16(3): 9.
WANG Q, OPPERMAN LA, HAVILL LM, CARLSON DS, DECHOW PC (2006) Inheritance of sutural pattern at the pterion in rhesus monkey skulls. Anat Rec A Discov Mol Cell Evol Biol, 288(10): 1042-1049.
WILLIAMS L, BANNISTER L, BERRY M, COLLINS P, DYSON M, DUSSEK E (1998) Gray’s Anatomy. 38th ed. Churchill Livingstone, London, npp 568-595.
ZALAWADIA A, VADGAMA J, RUPARELIA S, PATEL S, RATHOD SP, PATEL SV (2010) Morphometric study of pterion in dry skull Gujarat Region. Njirm, 1: 25-29.